Epidemiology of drug resistance in the bacterium and identification of resistance and virulence factors.

1.Research interests:
1) Design and synthesis of novel chemicals entitles with potential biological activity for
therapeutic applications.
2) Developing polymer derivatives for drug delivery and biomedical implants.
3) Investigate the properties of plant derived essential oils and explore their therapeutic
potential.

Research Philosophy
Our projects are generally on the organic chemistry/biology interface. We are interested in
synthesis of natural product, Synthesis of 3-Aminoimidazo[1,2-a]Pyridine/pyrazine
derivatives, and extracting phytochemical using different solvents. These interests inevitably
take us towards designing novel substrates for inhibiting enzymes (medicinal chemistry).
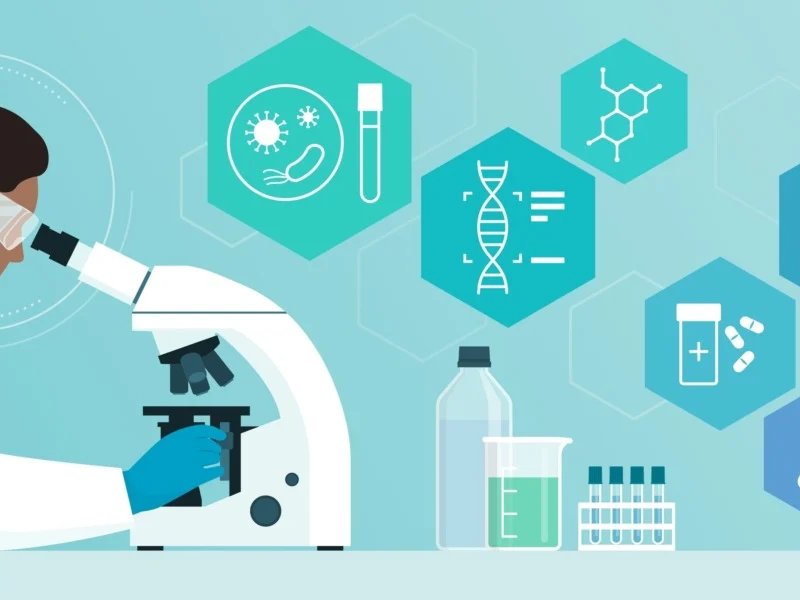
Synthesis of fluorinated isoflavones
Isoflavones, non-steroidal estrogens, are derived from leguminous plants such as soybeans and mung
beans. Isoflavones provide protection against estrogen-related ailments, including breast cancer,
osteoporosis, diabetes, and cardiovascular disease. Post-glycosylation, isoflavones exhibit low
aqueous solubility, diminished absorption, and rapid metabolism, hindering their ability to traverse
intestinal and hepatic barriers. The incorporation of fluorine atoms in a selective manner into naturally
occurring or biologically active structures typically enhances the biological profile. Fluorine's
diminutive size, strong electron affinity, and ability to form an additional hydrogen bond in receptors
render it an attractive candidate for drug design. Fluorine can alter the chemical, physical, and
biological properties of biologically active compounds, rendering it significant in pharmacology.
Fluorine, as a hydrogen bioisostere, enhances the lipophilicity of organic molecules and reduces their
basicity, facilitating cellular penetration and transport to active sites. The enhancement of fluorine's
absorption increases the oral bioavailability of medications. Two examples of isoflavone derivatives
synthesised in our lab showed remarkable anticancer activity.
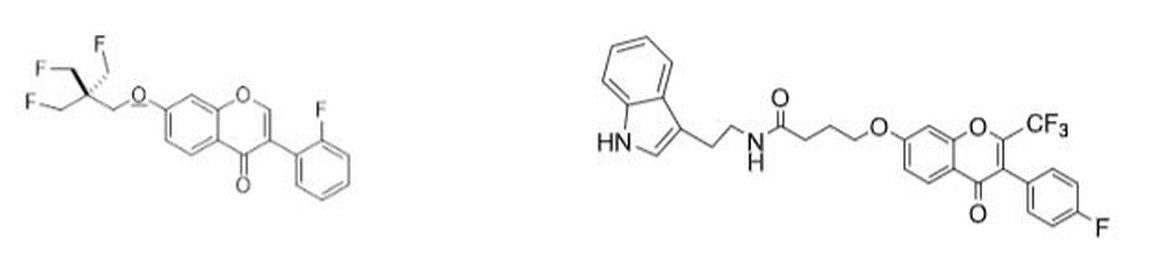
Synthesis of 3-Aminoimidazo[1,2-a]Pyridine/pyrazine Analogues
Imidazo[1,2-a]pyridines/pyrazines have recently become a highly relevant class of compounds in
medicinal chemistry and pharmaceutical research because of their exceptional adaptability and
therapeutic potential, which includes anticancer, antiprotozoal, antiviral, antimicrobial, and numerous
other bioactivities. Apart from conventional synthetic methods, novel approaches to creating
derivatives with structural diversity at the 2- and 3-positions of the imidazopyridine moiety are being
developed quickly to take advantage of the increasing interest in imidazo[1,2-a]pyridine properties.
Multicomponent reactions (MCRs) are thought to be capable of producing about 5% of the present
global pharmaceutical supply. The Groebke-Blackburn-Bienaymé multicomponent reaction (GBB-
3CR), a variation of these MCRs, synthesizes imidazo[1,2-a]pyridines effectively and economically.
In the one-pot, one-step GBB reaction, 2-aminoazines condense with catalytically activated aldehydes
and isocyanides to form imidazopyridine.
Extracting Essential oils from plants
Medicinal plants are nature gift to humanity, facilitating a disease prevention and a healthy
existence. Aromatic and medicinal plants are vital sources of secondary metabolites, which
have extensive applications in the management of plant and human illnesses, as well as in
the pharmaceutical, culinary, and cosmetic sectors. Essential oils (EOs) represent a diverse
and enduring category of medicinal plant preparations, derived from more than 17,500
aromatic species and contained within various plant structures, including fruits, flowers,
leaves, stems, and roots. Essential oils consist of a variety of molecular types across
different structural categories, including hydrocarbons—specifically mono- and
sesquiterpene hydrocarbons—as well as oxygenated mono- and sesquiterpenes featuring
various functional groups, such as aldehydes, alcohols, ketones, esters, epoxides, and
volatile phenolics. Numerous studies have demonstrated that medicinal plants are a major
source of bioactive phytochemicals with antibacterial capabilities. These molecules have the
ability to protect the human body from free radical-induced stress, which can cause cardiac
and neurological problems, joint inflammation, cancer, and other dysfunctions.
The Essential oils from plants collected from Palestine with special emphasis on Plants
growing in the unique place Jericho/Palestine, the lowest place on earth, are extracted via
steam- or hydrodistllation were identified using GC-MS. Additionally, Methanol, ethyl acetate
and dichloromethane extract are studied and their flavonoids and polyphenols content are
evaluated.
These projects also lead us towards designing inhibitors of pathways specific to bacteria and
fungi in order to generate novel antibiotics.
Scientific activities and grants:
Dr. Nisreen Alhaj was awarded the Scientific high level visiting fellowship (SSHN) for four cosecutive years from 2022-2025: The SSHN fellowship is funded by the French consulate in Jerusalem for one-month research visit at Molecular and Material Chemistry group - C2M, Bordeaux University-France in collaboration with Dr. Emilie Genin.
Dr. Nisreen Alhaj was awarded the 4th Palestinian Quebecer Science Bridge (PQSB) Call 2023 funded by Fonds de Recherche Du Quebec – Nature Et Technologies (FRQNT), Santé (FRQS), and Société Et Culture (FRQSC): three-month research visit at Génie desmines, de la métallurgie et des matériaux, Laval University, Canada in collboration with
Dr.Hendra Hermawan.
Links:
https://www.facebook.com/story.php/?story_fbid=510091641409223&id=100072251748120
https://www.facebook.com/share/p/1CJvs188gP/?mibextid=wwXIfr
Dr. Nisreen Alhaj received a position of a visiting associate professor for three years (2024-2027) at Génie des mines, dela métallurgie et des matériaux, Laval University,Canada. https://www.ulaval.ca/la-recherche/repertoire-corps-professoral/nisreen-alhaj
A.P., Murphy, C.D., Cormanich, R.A. and Hagan,
Cox JA, O'Hagan D.
Sawafta A, Hidmi A.